What are some current and prospective cures and treatments which use stem cells? What do they do? How exactly do they work? . . .
Treatments and cures which use stem cells and stem cell technologies have existed for many years. Numerous experiments, studies, clinical trials are currently underway for different types of stem cell therapies. Using stem cells to treat disease is a quintessence of translational medicine - bringing cures and treatments from the lab bench to the bedside. The following are some stem cell treatments and cures of today and tomorrow. | 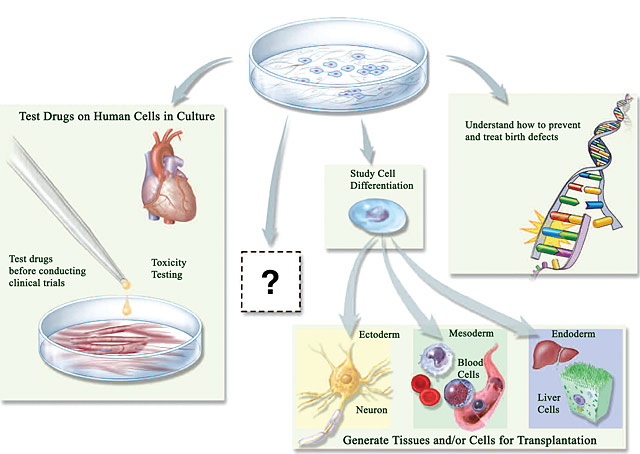 The use of stem cells for medical treatments is a fascinating example of translational research and its numerous benefits. (Image Citation 34) |
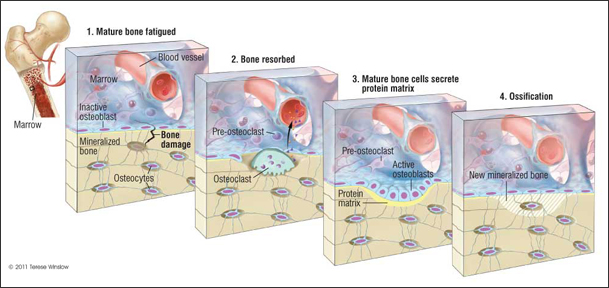
Building Bones With Stem Cells - Revolutionary Tissue Engineering Human bones, while often cited as some of the strongest structures in the human body, often require replacement in medicine. Diseases and conditions such as osteogenesis imperfecta (a type of brittle bone disease) and osteoporosis (weakening of bone) often call for new bone growth. With an "estimated 40 million persons in the United States [who currently] either have or are at risk for osteoporosis], new strategies are needed to promote new bone growth". Thus, stem cell research comes into play. For example, scaffolds ("implantable material, natural or synthetic, that provides appropriate support to a developing tissue") are necessary in tissue engineering. Ideally, scaffolds can enable stem cells to retain the ability to renew and differentiate, as well as "conform to the mechanical specifications of the injury site", ultimately being resorbed into the body "without generating toxic by-products". While many researchers have extensively investigated collagen, silk, chitosan, and other materials to use in scaffolds, they are limited in terms of purity and certain mechanical properties. Now, researchers are currently working on utilizing "bone marrow stromal cells (BMSCs)" (also known as mesenchymal stem cells or MSC) and differentiating them into osteoblasts (mature bone cells) to engineer bone tissue. MSCs offer both biocompatibility and durability as scaffolding material in osteogenic tissue engineering. Researchers have already explored the use of MSC for healing through secretion of molecules in inflammation and angiogenesis. MSCs can regenrate bone by "isolating the cells from the patient, culturing them to expand the cell number in vitro, seeding them onto scaffolds, and implanting the scaffold". Ultimately, MSC can be modified to proliferate at different rates and gain either tense cytoskeletons to become bone cells, or loose cytoskeletons to become fat cells. When implanted, MSCs thus can be coaxed to become new, invigorated bone cells. Studies have already demonstrated that "human detal and adult MSCs delivered in polymeric scaffolds can repair large bone defects in the rat". Furthermore, "limited studies have also shown that autologous MSCs seeded into shaped biomaterial scaffolds can be implanted to replace or repair hman bone defects. While this application of MSCs is currently in pre-clinical stages, and challenges (such as MSC isolation or unwanted epigenetic changes) exist, MSCs for bone regeneration hold promise of an innovative therapy. (Citation 42), (Citation 43), (Citation 44) |
|
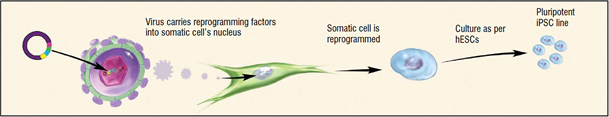
Using Induced Pluripotent Stem cells (iPSCs) in Medicine Induced Pluripotent Stem Cells have often been hailed by many researchers as a revolution in the field of stem cell research. Dr. Shinya Yamanaka, a researcher now affiliated with the Gladstone Institutes, created these stem cells in 2007. Yamanaka's work involved inserting four different transcription factors (now called Yamanaka factors: Oct 3/4, Sox2, Klf4, c-Myc) into an adult stem cell, giving it pluripotent properties (much like those of an embryonic stem cell). iPSCs have a large value for medicine. On NIH's "Stem Cell Information" Charles A. Goldthwaite, Jr., Ph.D. remarks in "The Promise of Induced Pluripotent Stem Cells (iPSCs)" that the novel reprogramming technology "offers the potential to treat many diseases, including Alzheimer's disease, Parkinson's disease, cardiovascular disease, diabetes, and amyotrophic lateral sclerosis (ALS; also known as Lou Gehrig's disease)". Because the cells used for generation of iPSCs would originate from the patient, "immune rejection of the differentiated derivatives would be minimized". Subsequently, "the need for immunosuppressive drugs to accompany the cell transplant would be lessened and perhaps eliminated altogether. iPSC therapies are already being created. In 2008, Dimos JT, et al. demonstrated that iPSCs derived from a patient with ALS could be directed to differentiate into motor neurons, which are the cells that are destroyed in the disease. In another experiment conducted by Zhang J, et al., Mauritz C, et al., Kuzemenkin A, et al., and Pfannkuche K, et al., showed that iPSCs "created from human and murine fibroblasts can give rise to functional cardiomyocytes that display hallmark cardiac action potentials". While these cells cannot be used for therapy yet because "cardiac development of iPSCs is delayed compared to that seen with cardiomyocytes derived from [embryonic stem cells] or fetal tissue", and "variation exists in the expression of genetic markers in the iPSC-derived cardiac cells as compared to that seen in [embryonic stem cell]-derived cardiomyocytes", researchers continue to study the iPSCs to cross these barriers to using these stem cells as actual treatments. (Citation 45), (Citation 46), (Citation 47), (Citation 48), (Citation 49) |
|
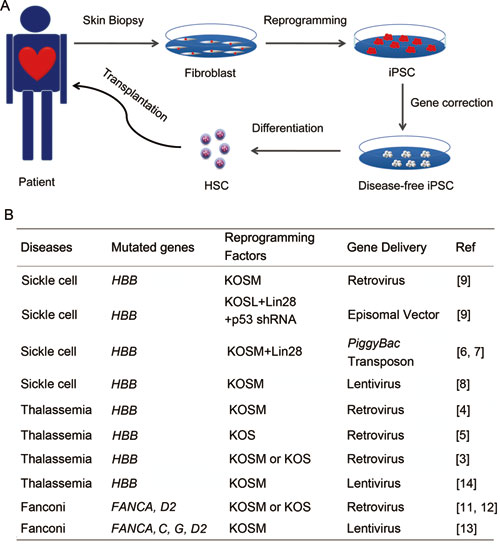
iPSC Based Gene Therapy for Inherited Red Blood Cell Disorders Red blood cell disorders are a particularly difficult-to-treat illness. According to Xu et al. (2012), until recently, "hematopoietic stem cell transplantation remains the only curative therapy for certain genetic diseases of the hematopoietic system, including some inheritable deficiencies in red blood cells". While the traditional treatment method of hematopoietic stem cell [HSC] transplantation "from Human Leukocyte Antigen (HLA)-matched donors or gene corrected autologous HSC" was "attractive and promising", obstacles such as difficulties in finding fully-matched donors, low efficiency rates for gene delivery, and safety concerns due to random viral vector integration into host genomes prevented this option of treatment from becoming ideal. However, now, iPSCs may be able to "provide a new promising strategy for cell therapy of RBC diseases". iPCs show an "unlimited proliferative ability" in culture, and are "amenable to gene editing" for correction of the disease mutations. The researchers report that iPSCs, with new and innovative technologies like lineage conversion, "may constitute [...] a valid alternative to HSCs for the treatment of anemic patients" in the future. Xu and colleagues tested iPSCs in β-thalassemia (a progressive anemia caused by the deficiency of β-global chain (HBB) expression during fetal to adult transition, which is common worldwide) patients. Patient iPSCs "were transduced with viral vectors expressing β-globin at a low multiplicity of infection (MOI), and then iPSC lines harboring a single copy of β-globin from a single viral integration were isolated". 15 of the iPSC lines with single viral integrations "were randomly picked up, only one of which met all five criteria" which Papapetrou et al. identified as to "reduce oncogenic potential of genome-integrated exogenous transgenes". When differentiated into erythrocytes, "the expression of vector-encoded β-globin reached about 85% of normal β-globin levels". While these erythrocytes do not yet express the "adult β-globin", researchers continue to explore the potential of iPSCs in β-thalassemia. (Citation 50), (Citation 51) |
|
"The Skin Gun" - An Innovative Stem Cell Treatment for Burns In the world of developing biomedical sciences, rarely does a cure or treatment come forth with such promising results as the "The Skin Gun". Created by Jörg C. Gerlach, MD, PhD, a Professor of Surgery and Bioengineering at the University of Pittsburgh, the "skin gun" uses stem cells to regenerate patient skin from severe burn injuries by using the patient's own stem cells. According to an article from the McGowan Institute for Regenerative Medicine, surgeons take a sample from a healthy piece of skin and isolate skin cells, including skin stem cells, using a method Dr. Gerlach and his colleagues developed. "Then the skin gun comes into play. A surgeon loads the stem cells into a sterile syringe, loads the syringe into the nozzle like a cartridge, and sprays the cells through the nozzle directly onto the wound". This stem cell therapy holds great promise for effective burn injury treatment.
| A demonstration of Dr. Gerlach's "Skin Gun", currently in development and clinical trial phases. (Video Citation 8) |